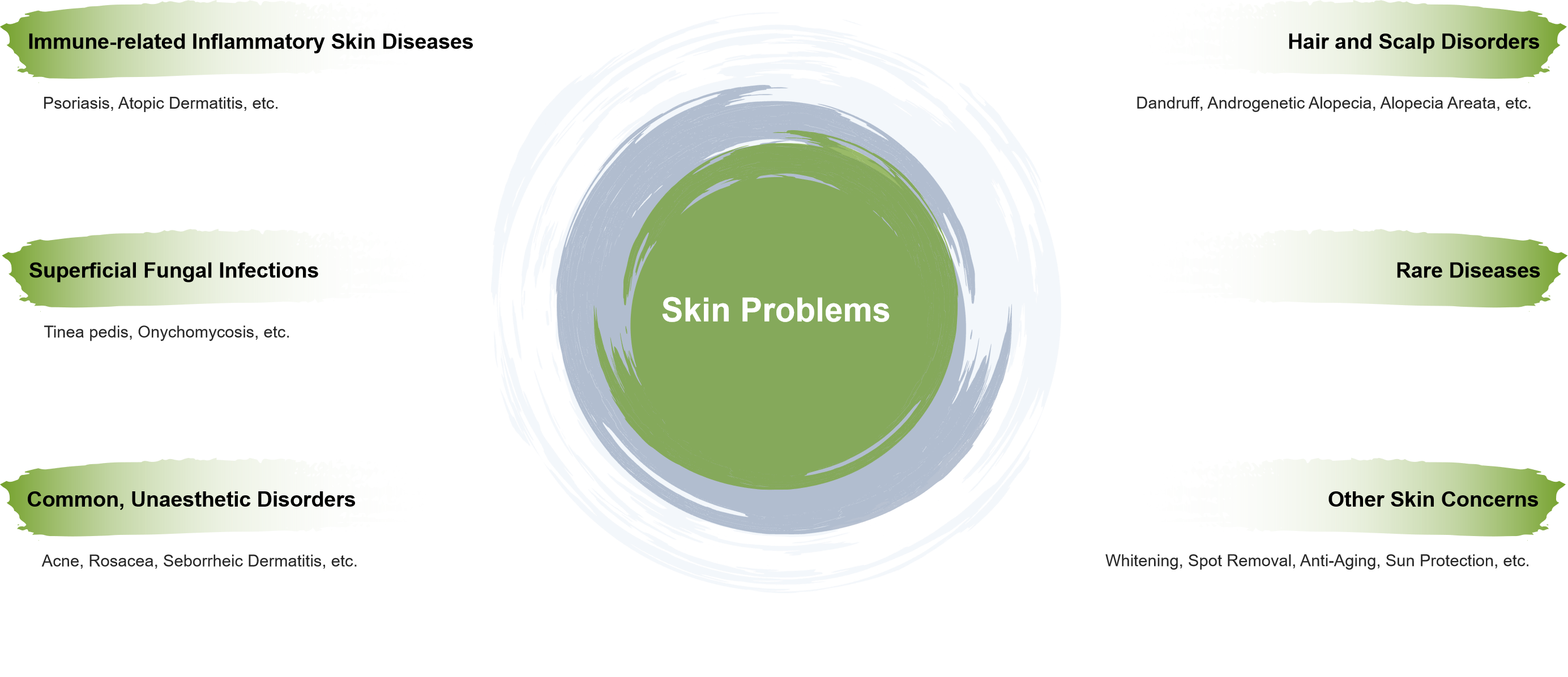
Sinomune Pharmaceutical Co., Ltd. (江苏知原药业股份有限公司) is a leading dermatological pharmaceutical company, specializing in the R&D, manufacturing, marketing, and sales of innovative and generic medicines, medical devices, and cosmetics in China.
Our innovative "Online + Offline" business model, combined with our market-leading brands in acne and rosacea (丽芙/LIFU), topical anti-fungal treatments (洛芙/LUOFU), and topical psoriasis treatments (金纽尔/JINUER), establishes us as a key player in the Chinese dermatology market.

With a a state-of-the-art, fully automated production system, our current topical formulation manufacturing capacity reaches 200 million units (tubes/bottles) annually, and plan to scale annual output to 350 million units within the next three years.
With16 topical production lines in operation, we can produce a diverse range of topical formulations, including creams, ointments, gels, solutions, lotions, and nail lacquers. We also have OEB high-activity workshops and explosion-proof workshops.

License-In or Co-Development:
1. Innovative drugs, medical devices, skin diagnosis, test & technology platforms
2. Overseas dermatological medications with distinct advantages
3. Efficacy-proven functional skincare products, special cosmetic ingredients.
We Also Provide:
1. Out-license opportunity of our products
2. CDMO/CMO services, including locolization of innovative topical drugs.
Contact us:BD@sinomune.com
News:
Sinomune Pharmaceuticals Strengthens Dermatology Portfolio with Exclusive License for Innovative Acne Treatment Sarecycline in China from Almirall.
Wuxi, China & Barcelona, Spain –
Sinomune Pharmaceutical Co., Ltd. ("Sinomune"), a leading dermatology-focused pharmaceutical company in China announced they have entered into an exclusive license and distribution agreement for sarecycline in Mainland China, Hong Kong, and Macau with Almirall, S.A. ("Almirall"), a global biopharmaceutical company dedicated to medical dermatology,.
Under the terms of the agreement, Sinomune acquires the exclusive rights to commercialize sarecycline in the designated territory, a novel, once-daily narrow-spectrum oral antibiotic for acne with anti-inflammatory properties and better tolerability profile than classic tetracyclines, that puts an end to decades of lack of innovation in the field of systemic antibiotics for acne. This collaboration leverages Sinomune's extensive commercial footprint and expertise in the Chinese dermatology market alongside Almirall's innovative portfolio and commitment to addressing the needs of people living with skin diseases in different populations around the world.
Sarecycline represents an important addition to Sinomune's dermatology portfolio, reinforcing the company's commitment to providing patients and dermatologists with advanced therapeutic options that address significant unmet medical needs. The product is indicated for the treatment of inflammatory lesions of non-nodular moderate to severe acne vulgaris in patients 9 years of age and older.
"Sarecycline is an innovative treatment with a compelling clinical profile for patients with moderate to severe acne," said a spokesperson for Sinomune. "Securing the exclusive rights to this asset aligns perfectly with our mission to bring meaningful, differentiated treatments to the Chinese market and strengthens our leadership in dermatology."
Approved by the U.S. FDA in 2018 under the brand name Seysara® and recently by China’s China’s National Medical Products Administration (NMPA) in 2025, sarecycline is a tetracycline-derived antibiotic. Its efficacy and safety have been demonstrated in over 2,300 subjects aged 9 years and older across three multicenter, randomized, double-blind, placebo-controlled Phase 3 clinical studies. The product shows reductions from baseline in inflammatory lesion counts and improvements in the percentage of patients with cleared or almost cleared facial lesions based on an Investigator’s Global Assessment (IGA); these effects were observed, as early as week 3 and maintained through week 12.
About Sinomune Pharmaceutical Co., Ltd.
Sinomune Pharmaceutical Co., Ltd. is a leading dermatology-focused pharmaceutical company in China, with over thirty years of dedication to the skin health sector. Recognized as a "National-level Little Giant Enterprise", Sinomune boasts a strong product portfolio including LuoFu (洛芙®), LiFu (丽芙®), JinNiuer (金纽尔®), and Zrvive (质润®), which are dedicated to addressing conditions such as fungal infections, acne, inflammatory skin diseases, and scalp disorders.
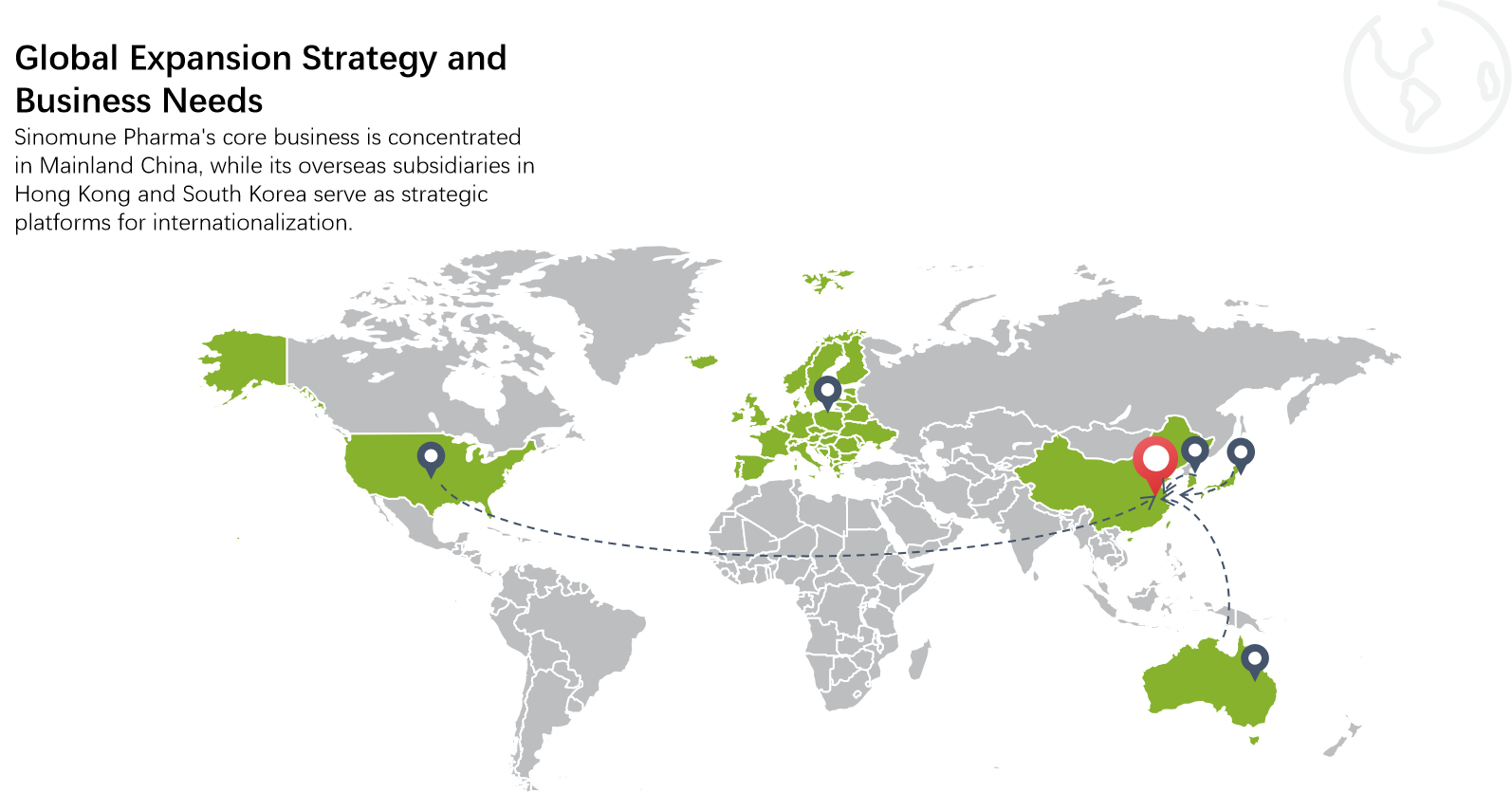
Sinomune operates a national postdoctoral research station, a CNAS-certified laboratory, and several provincial R&D platforms. Moving forward, Sinomune will continue to advance its "Technology + Marketing" dual-drive strategy, expand its product coverage, enrich its product lines, accelerate global expansion, and strive to be a leader in the dermatology field.
About Almirall
Almirall is a global biopharmaceutical company dedicated to medical dermatology. We closely collaborate with leading scientists, healthcare professionals, and patients to deliver our purpose: to transform the patients' world by helping them realize their hopes and dreams for a healthy life. We are at the forefront of science to deliver ground-breaking, differentiated medical dermatology innovations that address patients´ needs.
Almirall, founded in 1944 and headquartered in Barcelona, is publicly traded on the Spanish Stock Exchange (ticker: ALM, total revenue in 2024: €990 MM, over 2000 employees globally). Almirall products help to improve the lives of patients every day and are available in over 100 countries.
1. Pariser DM, Green LJ, Lain EL, Schmitz C, Chinigo AS, McNamee B, Berk DR. Safety and Tolerability of Sarecycline for the Treatment of Acne Vulgaris: Results from a Phase III, Multicenter, Open-Label Study and a Phase I Phototoxicity Study. J Clin Aesthet Dermatol. 2019 Nov;12(11):E53-E62. Epub 2019 Nov 1. PMID: 32038757; PMCID: PMC6937166.